 Новости отрасли
Новости отрасли Анализ мировой индустрии лабораторных приборов и оборудования: состояние рынка, региональная структура и конкурентная модель в 2026 году.
Анализ мировой индустрии лабораторных приборов и оборудования: состояние рынка, региональная структура и конкурентная модель в 2026 году.
2026-03-16
2026-03-12
2026-03-10
2026-02-27
2025-11-20
2025-11-20
 Текущие события
Текущие событияIntroduction
The landscape of metabolic disorder treatments has been revolutionized by the introduction of novel peptide therapeutics. Among these breakthroughs, Tirzepatide stands out as a highly effective, next-generation molecule. For pharmaceutical procurement managers and R&D scientists, sourcing a highly purified, strictly compliant Tirzepatide active pharmaceutical ingredient (API) is the foundational step in drug development and generic formulation. This article provides an in-depth overview of Tirzepatide's chemical profile, quality specifications, and clinical applications, ensuring your development pipeline is supported by the highest industry standards.
| Item | Content |
| English Name | Tirzepatide |
| English Synonyms | Tirzepatide;GIP\GLP-1;Tirzepatide (LY3298176);Trizepatide;Gip\GLP-1 Tirzepatide Ly3298176 Peptide;Tilposide;Tirzepatide(GLP-1);terzapitide |
| CAS No. | 2023788-19-2 |
| Molecular Formula | C225H348N48O68 |
| Molecular Weight | 4813 |
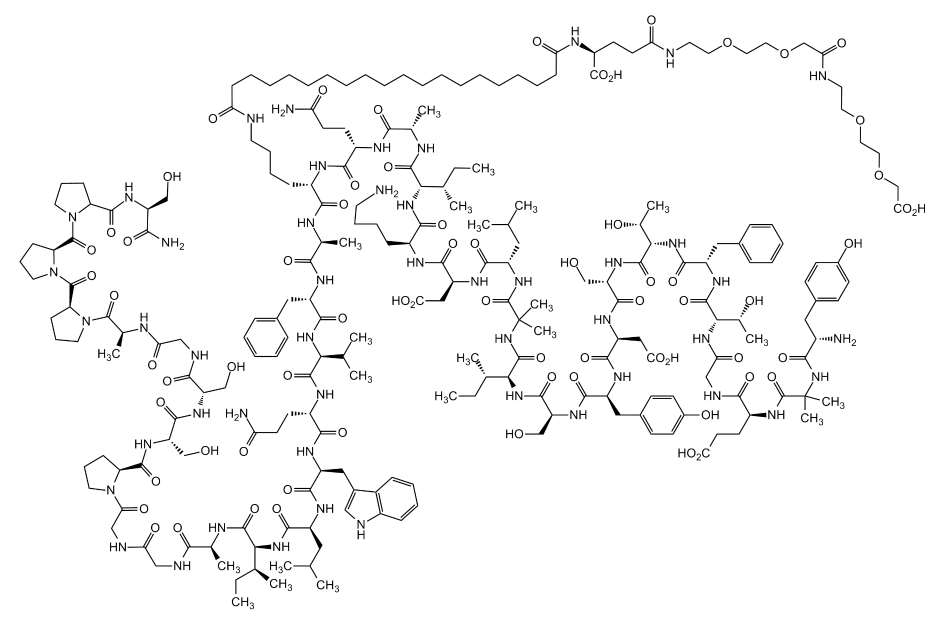
Tirzepatide is a pioneering, 39-amino acid synthetic peptide that functions as a dual agonist for both the glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptors. Recognized universally by its registry number, CAS 2023788-19-2, this molecule is engineered with a unique C20 fatty diacid moiety attached via a hydrophilic linker.
This specific structural modification allows the peptide to bind tightly to plasma albumin in the bloodstream, significantly extending its half-life to approximately five days and enabling convenient once-weekly subcutaneous dosing.
Chemical Formula: C225H348N48O68
Molecular Weight: 4813.45 g/mol
For laboratories and pharmaceutical manufacturers aiming to scale their production, securing a highly verified CAS 2023788-19-2 Tirzepatide GMP Peptide API ensures that the raw material meets the rigorous safety, efficacy, and consistency thresholds mandated by international regulatory bodies such as the FDA and EMA.
Appearance: White to off-white lyophilized solid powder.
Purity (HPLC): ≥ 99.0%, ensuring minimal peptide-related impurities and truncated sequences.
Peptide Content (N%): ≥ 80.0%, confirming the high concentration of the active molecule.
Water Content (Karl Fischer): ≤ 5.0%, maintaining optimal stability during long-term storage.
Endotoxin Limit: ≤ 10 EU/mg, a critical and strictly monitored parameter making it the ideal Tirzepatide Injectable API For Diabetes Clinical R&D.
Residual Solvents: Compliant with ICH Q3C guidelines.
Tirzepatide's dual-receptor mechanism offers a synergistic effect that goes beyond traditional GLP-1 mono-agonists. By engaging both GIP and GLP-1 pathways, it enhances insulin secretion, significantly suppresses glucagon production, and delays gastric emptying, leading to exceptional clinical outcomes.
Type 2 Diabetes Mellitus (T2DM) Management: Tirzepatide is clinically proven to lower HbA1c levels more effectively than many existing therapies. It improves insulin sensitivity and preserves pancreatic beta-cell function, making it a cornerstone for modern anti-diabetic formulations.
Obesity and Chronic Weight Management: The medical community widely recognizes the Tirzepatide Dual GIP GLP-1 Agonist For Weight Loss as a generational breakthrough. It promotes profound satiety and reduces food intake, leading to unprecedented weight reduction in comprehensive clinical trials.
Advanced Clinical R&D: Beyond diabetes and weight management, current global research is exploring Tirzepatide's potential in treating Non-Alcoholic Steatohepatitis (NASH), obstructive sleep apnea, and cardiovascular risk reduction. Our high-purity API supports innovators exploring novel delivery systems, such as oral peptide formulations or advanced auto-injector pens.
Q1: What are the required storage conditions for Tirzepatide API?
To preserve its molecular stability and prevent degradation, the lyophilized peptide powder should be stored at -20°C for long-term storage. For short-term transit or laboratory handling, temperatures between 2°C and 8°C are acceptable. It is imperative to protect the API from direct light and moisture.
Q2: Is your facility capable of supporting large-scale commercial manufacturing?
Yes. Our manufacturing facilities are fully GMP-compliant and equipped to scale seamlessly. We support everything from milligram-level synthesis for early-stage in-vitro studies to multi-kilogram commercial production for global market supply.
Q3: Do you provide regulatory support documentation for IND/ANDA submissions?
Absolutely. We understand the regulatory hurdles pharmaceutical companies face. We provide comprehensive documentation, including full Drug Master Files (DMF), Route of Synthesis (ROS) details, impurity profiles, and long-term stability study data to accelerate your regulatory submissions.
Disclaimer: This website respects intellectual property rights. If any infringement is found, please contact this website in a timely manner for handling.